Proteomics in Drug Discovery: A Revolution in Pharma
October 20, 2023I. Introduction
A. Definition and Overview of Proteomics
Proteomics is the large-scale study of proteins, particularly their structures and functions. Proteins are vital parts of living organisms, as they are the main components of the physiological metabolic pathways of cells. The term “proteomics” is a blend of “protein” and “genome,” and it refers to the entire set of proteins produced or modified by an organism. While genomics deals with the study of genes and DNA, proteomics addresses the products that these genes code for – the proteins.
B. Significance of Proteomics in Drug Discovery
Proteomics has an immense significance in the realm of drug discovery. Since proteins play crucial roles in virtually all biological processes, understanding protein structure, function, and interactions can lead to the identification of novel drug targets. By analyzing the proteome of a disease state and comparing it to a healthy state, researchers can pinpoint proteins that may be implicated in disease processes. These proteins can then be targeted for therapeutic intervention. Furthermore, proteomics can also assist in understanding drug mechanisms, predicting potential side effects, and tailoring drugs to individual patient profiles, leading to the rise of personalized medicine.
C. Objectives and Scope of the Presentation
This presentation aims to provide an in-depth exploration of proteomics, from its foundational concepts to its pivotal role in modern drug discovery. We will delve into the techniques employed in proteomics, illustrate its applications, and demonstrate its significance in designing next-generation drugs. Furthermore, the presentation will highlight the potential of proteomics in advancing personalized medicine and addressing the challenges faced in today’s drug discovery pipeline.
II. Basics of Proteomics
A. Definition and Purpose of Proteomics
Proteomics, at its core, is the systematic study of the entire set of proteins present in a particular cell, tissue, or organism at a specific time. Its purpose is to offer a comprehensive view of how proteins interact, function, and regulate cellular processes. By understanding the dynamic nature of proteins in health and disease, researchers can gain invaluable insights into disease mechanisms, discover potential biomarkers, and identify novel therapeutic targets.
B. Key Components of Proteomics
1. Protein Identification
This involves determining which proteins are present in a sample. Traditional methods might use antibodies or gel-based separations. Modern techniques, especially mass spectrometry, have made high-throughput protein identification feasible, allowing for the identification of thousands of proteins in a single experiment.
2. Protein Quantification
After identifying the proteins, it’s important to determine how much of each protein is present. This can give insights into how certain conditions, treatments, or mutations might upregulate or downregulate protein levels.
3. Protein Characterization
Beyond just knowing which proteins are present and in what quantities, it’s essential to understand their functional roles, post-translational modifications, interactions with other molecules, and three-dimensional structures.
C. Techniques in Proteomics
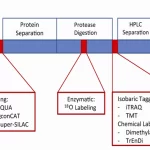
1. Mass Spectrometry
This is a pivotal tool in proteomics. Proteins are digested into smaller peptides, which are then ionized and measured based on their mass-to-charge ratio. Mass spectrometry can identify proteins, quantify their abundance, and even detect post-translational modifications.
2. Two-Dimensional Gel Electrophoresis (2D-PAGE)
A technique that separates proteins based on two properties: their isoelectric point (pI) and molecular weight. Proteins are first separated horizontally by pI and then vertically by size. This results in a 2D pattern, with each spot representing a unique protein or protein form.
3. Protein Microarrays
These are akin to DNA microarrays but are used to study proteins. They can help in studying protein-protein, protein-DNA, and protein-ligand interactions. Protein microarrays can also be instrumental in detecting antibodies in patient samples, thus aiding in disease diagnosis.
D. Proteomic Databases and Resources
As proteomic research advances, there’s a growing need for centralized databases to store, retrieve, and analyze protein data. Databases like UniProt, Protein Data Bank (PDB), and PRIDE are invaluable resources for researchers. They store information on protein sequences, structures, functions, and interactions. Additionally, tools like BLAST enable sequence comparisons, while software suites like MaxQuant or Skyline aid in the analysis of mass spectrometry data. Access to these databases and tools empowers researchers to build on existing knowledge, fostering collaborative and progressive research in the field.
III. Role of Proteomics in Drug Discovery
A. Target Identification and Validation
1. Identifying Potential Drug Targets
The comprehensive profiling of proteins in a disease state versus a healthy state can unveil proteins that are either upregulated, downregulated, or modified. Such proteins can be potential drug targets. Once identified, these targets are then subject to further studies to validate their importance in the disease mechanism.
2. Validating Target Proteins
Not all proteins identified as potential targets are suitable for drug discovery. Validation involves a series of experimental studies to ascertain the significance of a protein in disease progression and its potential as a therapeutic target. This step is crucial, as it ensures that time and resources are invested in targets with the highest potential for therapeutic intervention.
B. Biomarker Discovery
1. Biomarkers in Disease Diagnosis
Biomarkers are molecules that indicate the presence or severity of a disease. By comparing the proteomic profiles of healthy and diseased states, researchers can identify proteins that serve as potential biomarkers. These biomarkers can assist in early disease detection, prognosis, and monitoring treatment efficacy.
2. Biomarkers for Drug Response Prediction
Certain proteins can indicate how a patient might respond to a particular drug. Identifying such biomarkers can help in selecting the right drug for a patient, ensuring better treatment outcomes and reducing adverse reactions.
C. Drug Mode of Action Studies
1. Understanding How Drugs Interact with Proteins
Proteomics can reveal the specific proteins a drug binds to and how this interaction affects the protein’s function. This insight can be invaluable in understanding the drug’s therapeutic mechanism.
2. Uncovering Off-Target Effects
No drug is entirely specific to its target. Proteomics can help identify unintended protein targets of a drug, giving insights into potential side effects or additional therapeutic uses.
D. Toxicology and Safety Assessment
1. Evaluating Drug-Related Toxicity
By studying changes in protein expression or modification in response to drug exposure, researchers can detect early signs of toxicity and potential harm to organs or cellular processes.
2. Predicting Adverse Effects
Comparing the proteomic profiles before and after drug administration can hint at potential adverse effects, enabling researchers to modify drug design or dosage for optimal safety.
E. Personalized Medicine
1. Tailoring Drug Treatments Based on Proteomic Profiles
Everyone is unique, and so is our proteomic profile. By analyzing an individual’s proteome, treatments can be tailored to ensure maximum efficacy and minimal side effects.
2. Case Studies in Personalized Medicine
There have been instances where proteomic information has revolutionized treatment approaches. For example, the identification of specific protein mutations in certain cancers has led to the development and use of targeted therapies, significantly improving patient outcomes.
In conclusion, proteomics plays a vital role across the spectrum of drug discovery, from identifying targets to tailoring treatments for individual patients. As our understanding and technologies advance, proteomics is set to further revolutionize the world of therapeutics.
IV. Challenges and Limitations
A. Sample Preparation and Data Quality
Sample preparation is a critical step in proteomics, and minor inconsistencies can introduce significant errors or biases. Challenges include:
- Sample Heterogeneity: Tissues or cells may contain a diverse range of proteins, some of which might be in very low abundance and can be easily overshadowed by highly abundant proteins.
- Degradation: Proteins can degrade quickly, which can affect the accuracy of the proteomic analysis.
- Contamination: The risk of sample contamination from external proteins can interfere with results.
B. Data Analysis and Interpretation
Given the vast amount of data generated in proteomic studies, data analysis can be quite challenging:
- Complexity of Data: Mass spectrometry, for example, generates massive datasets that require specialized software for analysis.
- False Positives: Given the high-throughput nature of proteomic experiments, there’s a risk of identifying proteins that aren’t genuinely related to the condition under investigation.
- Inter-laboratory Variability: Different labs might get slightly different proteomic profiles for the same sample due to variations in methodologies, equipment, or data analysis techniques.
C. Cost and Technology Accessibility
Advanced proteomic techniques can be expensive:
- High Equipment Cost: Instruments like mass spectrometers are costly to purchase and maintain.
- Training: Adequate training is required to operate sophisticated proteomic tools, which can add to the costs.
- Accessibility: Not all research institutions or countries have equal access to the latest proteomic technologies, which can hinder global collaboration or lead to disparities in research quality.
D. Ethical Considerations in Proteomics Research
As with all scientific research, ethical considerations play a significant role:
- Data Privacy: Proteomic data can potentially be used to identify individuals, posing risks to privacy.
- Consent: Particularly when studying human samples, informed consent must be obtained, and the use of samples must adhere strictly to the conditions under which that consent was given.
- Animal Testing: Proteomic studies often involve animal models, and there are ethical concerns regarding their use, care, and treatment.
In sum, while proteomics offers a wealth of opportunities to advance our understanding of biology and medicine, researchers must navigate a range of challenges and limitations. By acknowledging and addressing these challenges, the field can continue to grow and contribute valuable insights into health and disease.
V. Recent Advances in Proteomics
A. Advances in Mass Spectrometry
Mass spectrometry (MS) has been at the forefront of proteomics, and its continuous evolution has driven many advances in the field.
- Higher Resolution Instruments: The development of newer MS instruments with higher resolution and accuracy allows for more precise protein identification and quantification.
- Tandem MS (MS/MS): This technique, which involves multiple rounds of mass spectrometry, provides detailed information on peptide sequences and post-translational modifications.
- Ambient Mass Spectrometry: Techniques like DESI (Desorption Electrospray Ionization) allow for direct sampling from surfaces without extensive sample preparation.
B. Integration with Genomics and Transcriptomics
With the rise of multi-omics approaches, integrating proteomic data with genomic and transcriptomic information offers a more holistic view of cellular function.
- Proteogenomics: This involves using proteomic data to annotate and validate gene models, providing a feedback loop that enhances both genomic and proteomic analyses.
- Correlation Studies: By comparing transcriptomic and proteomic data, researchers can gain insights into post-transcriptional regulation mechanisms.
C. Single-Cell Proteomics
While traditional proteomics typically analyzes bulk samples containing many cells, recent advances have focused on the proteomes of individual cells.
- Enhanced Sensitivity: The development of highly sensitive MS techniques has made it feasible to study protein expression in single cells.
- Heterogeneity Analysis: Single-cell proteomics can reveal cellular heterogeneity within tissues or tumors, providing insights into cell-to-cell variability in health and disease.
D. AI and Machine Learning in Proteomics
Artificial intelligence (AI) and machine learning (ML) are revolutionizing data analysis in proteomics.
- Data Mining: AI algorithms can sift through vast proteomic datasets to uncover patterns or biomarkers that might be missed by traditional analysis.
- Predictive Modeling: ML can be used to predict protein structures, interactions, or even the potential effects of drugs on protein networks.
- Integration with Other Data Types: AI can assist in integrating proteomic data with other omics data, enhancing the insights derived from multi-omics studies.
The pace of innovation in proteomics is rapid, and these advances are continually expanding the horizons of what’s possible in the field. The integration of cutting-edge technologies and interdisciplinary collaboration promises to bring even more groundbreaking discoveries in the near future.
VI. Case Studies and Success Stories
A. Examples of Drugs Developed Using Proteomics
1. Trastuzumab (Herceptin): Developed for breast cancer patients with tumors overexpressing the HER2 protein. Proteomic studies identified an overabundance of HER2 in certain breast cancer subtypes, leading to the development of this targeted therapy.
2. Imatinib (Gleevec): A breakthrough in the treatment of chronic myeloid leukemia (CML). Proteomic studies helped understand the BCR-ABL fusion protein’s role, a tyrosine kinase. Gleevec specifically inhibits this kinase, revolutionizing CML treatment.
B. Biomarker Discovery Success Stories
1. Prostate-Specific Antigen (PSA): Though known before the rise of modern proteomics, it’s a classic example of a protein biomarker used in prostate cancer screening. Continuous proteomic research aims to refine the use of PSA and identify new, more specific biomarkers.
2. Cardiovascular Diseases: Proteomic approaches have identified potential biomarkers like troponins and natriuretic peptides, which are now instrumental in diagnosing and managing heart-related conditions.
C. Personalized Medicine Implementation Cases
1. Cystic Fibrosis and CFTR Modulators: Through the integration of genomics and proteomics, researchers identified different mutations in the CFTR gene responsible for cystic fibrosis. This has led to the development of personalized treatments, like Ivacaftor, which are designed to address specific mutations.
2. Oncotype DX: This diagnostic test evaluates the expression levels of a set of genes in breast cancer tumors, providing insights into the likely course of the disease and the potential benefit of chemotherapy. By integrating genomic and proteomic data, this test exemplifies personalized medicine by helping to tailor treatments to individual patients.
These case studies highlight the transformative impact of proteomics in drug discovery, diagnostics, and personalized medicine. By focusing on the intricate world of proteins, researchers have been able to develop more effective and tailored treatments, offering hope to countless patients worldwide.
VII. Future Directions and Trends
A. Emerging Technologies in Proteomics
1. Real-time Monitoring: Advanced sensors and devices are being developed to monitor protein levels and interactions in real-time, giving a dynamic view of cellular processes as they unfold.
2. CRISPR/Cas Systems: Though primarily a genomic tool, CRISPR is starting to find applications in proteomics, allowing researchers to target specific proteins or modifications for analysis.
3. Hyperplexed Proteomics: Novel labeling methods are enabling the simultaneous analysis of multiple samples, increasing throughput and comparative capabilities.
B. Multi-Omics Integration
1. Systems Biology Approaches: By integrating proteomic, genomic, metabolomic, and other data, a systems-level view of biology is emerging, leading to more holistic insights and therapeutic strategies.
2. Cloud Computing and Big Data: As multi-omics studies generate vast amounts of data, cloud-based platforms and advanced data analytics will be critical for data storage, sharing, and analysis.
C. Proteomics in Drug Repurposing
1. Off-target Effects: Proteomics can identify unintended protein interactions of existing drugs, which could hint at new therapeutic uses for already approved drugs.
2. Disease Mechanism Insights: By understanding the proteomic underpinnings of diseases, drugs initially developed for one condition might be repurposed for another with a similar proteomic profile.
D. Regulatory Considerations in Proteomics-Based Drug Development
1. Quality Control: As proteomic data starts to play a more prominent role in drug development and personalized medicine, there will be increased scrutiny on data quality, reproducibility, and standardization.
2. Ethical and Data Privacy Considerations: Proteomic data, especially when combined with other personal data, can raise privacy concerns. Regulatory bodies will likely develop guidelines to ensure data protection while not stifling innovation.
3. Accelerated Approvals: As proteomic insights lead to more targeted therapies with clear mechanistic rationales, we might see regulatory bodies offering accelerated approval pathways, especially for conditions with unmet medical needs.
In conclusion, the future of proteomics is incredibly promising. As technologies advance and integration with other scientific disciplines increases, proteomics will continue to reshape our understanding of biology and revolutionize therapeutic approaches. However, with these advancements come challenges that will require collaboration, innovation, and careful consideration to navigate effectively.
VIII. Conclusion
A. Recap of Key Points
1. Proteomic Landscape: Proteomics delves deep into the intricate world of proteins, providing insights into their roles, interactions, and modifications.
2. Drug Discovery: By identifying and validating drug targets, aiding in biomarker discovery, and elucidating drug modes of action, proteomics plays a pivotal role in drug discovery.
3. Challenges: While promising, proteomics faces challenges such as sample preparation, data analysis complexities, and ethical considerations.
4. Advances: Recent technological advancements, especially the integration of AI and machine learning, are propelling the field forward.
5. Case Studies: Examples like Herceptin and Gleevec underscore the tangible impact of proteomics in delivering transformative therapies.
B. Importance of Proteomics in Revolutionizing Pharma
Proteomics is not just another scientific discipline; it stands at the intersection of biology and medicine, holding the potential to revolutionize pharmaceuticals. By offering a deeper understanding of disease mechanisms, it facilitates the development of more targeted and effective therapies. Moreover, as personalized medicine becomes the gold standard, proteomics will be instrumental in tailoring treatments to individual patients, ensuring better outcomes and fewer side effects.
C. Future Impact and Potential
As we look to the future, the potential of proteomics seems boundless. With emerging technologies, it is set to delve deeper into cellular processes, uncovering mysteries that have eluded scientists for decades. Its synergy with other fields like genomics, metabolomics, and AI means that its impact will be amplified, leading to breakthroughs that were once deemed impossible.
In conclusion, proteomics, with its profound insights and technological innovations, stands poised to reshape the landscape of biomedicine. The coming years will undoubtedly witness its growing influence, with more success stories, novel therapies, and a deeper understanding of the very fabric of life. As researchers, professionals, and stakeholders in health, it’s an exciting journey we are embarking on, with proteomics lighting the way.