How are next generation sequencing technologies transforming genomic and genetic research?
November 24, 2023I. Introduction
A. Overview of Next-Generation Sequencing (NGS) Technologies
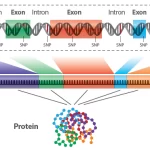
Next-Generation Sequencing (NGS) technologies represent a paradigm shift in genomic research, offering high-throughput sequencing capabilities that revolutionized the field. Unlike traditional Sanger sequencing, NGS enables the parallel sequencing of millions of DNA fragments, providing unprecedented speed and cost-effectiveness.
B. Significance in Genomic and Genetic Research
NGS has become a cornerstone in genomic and genetic research due to its ability to generate massive amounts of sequencing data rapidly. This technology has had a profound impact on various areas:
- Genome Sequencing: NGS allows the comprehensive sequencing of entire genomes, enabling the identification of genetic variations, rare mutations, and structural alterations.
- Transcriptomics: RNA sequencing (RNA-Seq) with NGS facilitates the quantification of gene expression, alternative splicing, and the discovery of novel transcripts, enhancing our understanding of gene regulation.
- Epigenomics: NGS is instrumental in studying epigenetic modifications, including DNA methylation and histone modifications, providing insights into gene regulation and cellular differentiation.
- Metagenomics: NGS enables the study of complex microbial communities in various environments, advancing our understanding of the human microbiome, environmental microbiology, and microbial ecology.
- Clinical Diagnostics: NGS is increasingly used in clinical settings for genetic testing, diagnosis of genetic disorders, and the identification of therapeutic targets, paving the way for personalized medicine.
C. Evolution from Sanger Sequencing to NGS
- Sanger Sequencing Era: Sanger sequencing, developed in the late 1970s, was the gold standard for DNA sequencing. While highly accurate, it was labor-intensive, time-consuming, and costly, limiting its scalability.
- NGS Revolution: The emergence of NGS technologies, starting with the 454 pyrosequencing and Illumina sequencing platforms, marked a transformative shift. NGS dramatically increased sequencing throughput and reduced costs, democratizing access to genomic data.
- Diversity of NGS Platforms: Over time, various NGS platforms, including Illumina, Ion Torrent, Pacific Biosciences (PacBio), and Oxford Nanopore, have been developed. Each platform has unique characteristics, such as read length, accuracy, and throughput, catering to diverse research needs.
- Ongoing Advancements: Continuous innovation in NGS technologies, such as improvements in read length, enhanced accuracy, and the development of long-read sequencing, has further expanded the applications and capabilities of genomic research.
The evolution from Sanger sequencing to NGS has propelled genomics into a data-rich era, fostering breakthroughs in understanding genetic diversity, unraveling disease mechanisms, and advancing personalized medicine.
II. Key NGS Technologies
A. Illumina Sequencing
- Short-read sequencing technology: Illumina sequencing is based on reversible dye terminator chemistry, producing short DNA fragments (reads). This technology is known for its high accuracy and throughput.
- Applications in whole-genome sequencing, exome sequencing, and RNA-seq: Illumina platforms are widely utilized for various applications, including whole-genome sequencing to identify genomic variants, exome sequencing to focus on protein-coding regions, and RNA-seq for transcriptome analysis.
B. Ion Torrent Sequencing
- Semiconductor-based sequencing: Ion Torrent sequencing relies on the detection of hydrogen ions released during DNA polymerization. It is a scalable and cost-effective approach for sequencing.
- Targeted sequencing and amplicon sequencing applications: Ion Torrent platforms are commonly employed for targeted sequencing, focusing on specific genomic regions of interest, as well as amplicon sequencing for studying targeted DNA fragments.
C. PacBio Sequencing
- Single Molecule Real-Time (SMRT) sequencing: Pacific Biosciences (PacBio) utilizes SMRT technology, enabling the observation of real-time DNA synthesis. This results in longer read lengths compared to short-read technologies.
- Long-read capabilities and applications in structural variant detection: PacBio sequencing excels in generating long reads, facilitating the detection of structural variants, complex genomic regions, and resolving repetitive sequences.
D. Nanopore Sequencing
- Real-time sequencing using nanopores: Nanopore sequencing involves threading DNA through nanoscale pores, and the changes in electrical current as the DNA passes through are used to decode the sequence in real-time.
- Long-read sequencing and portability: Nanopore sequencing offers long-read capabilities, making it valuable for de novo genome assembly and resolving complex genomic structures. Additionally, its portability is advantageous for field-based and point-of-care applications.
These key NGS technologies each have unique strengths, making them suitable for specific applications in genomic research, ranging from comprehensive genome analysis to targeted investigations of specific genomic regions.
III. Applications in Genomic Research
A. Whole-Genome Sequencing (WGS)
- Uncovering variations across the entire genome: Whole-genome sequencing involves analyzing the complete DNA sequence of an organism, enabling the identification of variations, including single nucleotide polymorphisms (SNPs) and structural variants, across the entire genome.
- Population studies and rare variant identification: WGS is instrumental in population genomics, providing insights into genetic diversity, population structure, and evolutionary relationships. It is also crucial for identifying rare variants associated with genetic disorders.
B. Exome Sequencing
- Focusing on protein-coding regions: Exome sequencing targets the protein-coding regions of the genome, known as the exome. This approach is cost-effective while capturing the majority of functionally relevant variants.
- Identifying disease-causing mutations: Exome sequencing is particularly valuable for identifying mutations responsible for Mendelian disorders and understanding the genetic basis of rare diseases.
C. RNA Sequencing (RNA-seq)
- Profiling gene expression and alternative splicing: RNA-seq provides a comprehensive view of the transcriptome by quantifying gene expression levels and detecting alternative splicing events. This enables the study of dynamic cellular processes.
- Transcriptomic studies and biomarker discovery: RNA-seq is applied in transcriptomic studies to understand gene regulation, discover novel transcripts, and identify potential biomarkers associated with diseases or specific conditions.
D. Epigenomic Studies
- DNA methylation and histone modification analysis: NGS technologies are employed in epigenomic studies to investigate modifications such as DNA methylation and histone modifications. These studies provide insights into epigenetic regulation and its role in normal development and diseases.
- Understanding epigenetic regulation in diseases: Examining epigenomic modifications helps unravel the role of epigenetics in various diseases, including cancer and neurological disorders. It contributes to understanding how epigenetic changes influence gene expression and cellular functions.
These applications showcase the versatility of NGS technologies in addressing diverse research questions across genomics, from characterizing the entire genome to deciphering the intricacies of gene expression and epigenetic regulation.
IV. Advancements in Genetic Research
A. Rare Variant Discovery
- Identifying rare genetic variations: Next-generation sequencing (NGS) technologies have significantly enhanced the ability to detect rare genetic variants. The identification of rare variants is crucial for understanding their role in diseases, especially rare genetic disorders.
- Implications in rare diseases and personalized medicine: The discovery of rare variants has direct implications for rare diseases, where a single genetic mutation can have a profound impact. Furthermore, in the context of personalized medicine, understanding rare variants contributes to tailoring treatments based on an individual’s unique genetic makeup.
B. Structural Variant Detection
- Uncovering large-scale genomic rearrangements: NGS technologies, particularly those with long-read capabilities like PacBio and Nanopore sequencing, excel in detecting structural variants. These include large-scale genomic rearrangements, duplications, deletions, inversions, and translocations.
- Impact on understanding genetic diversity: The identification of structural variants enhances our understanding of genetic diversity within populations. It provides insights into the genomic architecture and contributes to studies on population genetics and evolutionary processes.
C. Functional Genomics
- CRISPR-based functional genomics studies: The advent of CRISPR-Cas9 technology has revolutionized functional genomics. Researchers can now perform precise gene editing, allowing the investigation of gene function by creating targeted mutations and studying the resulting phenotypic changes.
- High-throughput screening and functional annotations: High-throughput techniques, enabled by NGS, facilitate large-scale functional genomics studies. These include screening approaches to assess the impact of genetic variations on cellular functions and the annotation of functional elements within the genome.
These advancements underscore the transformative impact of NGS technologies on genetic research, providing unprecedented insights into rare genetic variants, structural variations, and the functional understanding of the genome. This knowledge is foundational for advancing both basic science and clinical applications.
V. Data Analysis and Bioinformatics
A. Bioinformatic Pipelines
- Read mapping and variant calling: The analysis of NGS data involves bioinformatic pipelines that include crucial steps such as read mapping, aligning the sequenced reads to a reference genome, and variant calling, which identifies genomic variations. These pipelines are essential for accurately interpreting the wealth of data generated by NGS technologies.
- Integration with other omics data: Bioinformatic pipelines in NGS analysis often extend beyond variant calling. Integration with other omics data, such as transcriptomics or epigenomics, allows for a comprehensive understanding of the functional implications of genetic variations. This integrative approach enhances the biological insights derived from NGS experiments.
B. Challenges in NGS Data Analysis
- Big data challenges: The massive volume of data generated by NGS technologies poses significant big data challenges. Efficient storage, processing, and analysis of these large datasets require robust computational infrastructure and algorithms capable of handling the scale of genomic information produced.
- Quality control and standardization: Maintaining data quality and ensuring standardization across different NGS experiments are critical challenges. Quality control measures are essential to identify and address issues such as sequencing errors, artifacts, or biases, which can impact the accuracy of downstream analyses. Standardization efforts aim to establish consistent practices and benchmarks in NGS data analysis.
The bioinformatics aspect of NGS data analysis plays a pivotal role in translating raw sequencing data into meaningful biological insights. Addressing challenges in data analysis is key to harnessing the full potential of NGS technologies in genomics and genetic research.
VI. Impact on Precision Medicine
A. Genomic Profiling in Clinical Settings
- NGS applications in oncology: Next-Generation Sequencing (NGS) has revolutionized genomic profiling in clinical oncology. It enables comprehensive analysis of cancer genomes, facilitating the identification of specific genetic alterations associated with tumors. This detailed molecular characterization informs the development of targeted therapies and personalized treatment strategies.
- Tailoring treatment strategies based on genetic information: Genomic profiling through NGS allows clinicians to identify actionable genetic mutations and variations in individual patients. This information guides the selection of targeted therapies that specifically address the molecular characteristics of a patient’s cancer. The ability to tailor treatment strategies based on genetic insights enhances the precision and efficacy of cancer interventions.
B. Pharmacogenomics
- Personalized drug response prediction: NGS plays a crucial role in advancing pharmacogenomics, the study of how genetic variations influence individual responses to medications. By analyzing an individual’s genetic makeup, including variations in drug metabolism and response-related genes, NGS facilitates the prediction of personalized drug responses. This information helps clinicians optimize drug selection and dosage for better treatment outcomes.
- Improving drug efficacy and minimizing adverse reactions: Understanding the genetic factors that influence drug metabolism and efficacy allows for the optimization of medication regimens. NGS-based pharmacogenomic insights contribute to the avoidance of adverse drug reactions and the selection of drugs that are more likely to be effective for a particular patient. This personalized approach to drug therapy represents a significant advancement in precision medicine.
The impact of NGS on precision medicine extends to clinical applications, particularly in oncology and pharmacogenomics. By leveraging genomic information, NGS enhances the customization of medical interventions, leading to more effective treatments with reduced risks of adverse events.
VII. Emerging NGS Technologies and Innovations
A. Single-Cell Sequencing
- Analyzing individual cells for heterogeneity: Emerging NGS technologies, such as single-cell sequencing, enable the analysis of genetic information at the resolution of individual cells. This approach unveils cellular heterogeneity within tissues, allowing researchers to explore diverse cell populations and identify rare cell types. Single-cell sequencing provides insights into the genetic landscape of complex biological systems with unprecedented detail.
- Applications in cancer research and developmental biology: Single-cell sequencing has profound applications in cancer research, where intratumor heterogeneity can be dissected at the single-cell level. Understanding the genetic diversity within tumors informs targeted therapies and helps unravel mechanisms of resistance. In developmental biology, single-cell sequencing contributes to the exploration of embryonic development, tissue differentiation, and the identification of cell lineage trajectories.
B. Spatial Transcriptomics
- Visualizing gene expression in tissue context: Spatial transcriptomics is a cutting-edge NGS technology that integrates spatial information into gene expression profiles. By preserving the spatial context of RNA molecules within tissues, researchers can visualize where specific genes are expressed. This innovation allows for a more comprehensive understanding of cellular interactions within complex tissues.
- Advancements in understanding spatial relationships: Spatial transcriptomics goes beyond traditional RNA-seq by providing spatially resolved transcriptomic data. This advancement is particularly valuable in studying intricate biological processes, such as tissue development and disease pathology. Researchers can map gene expression patterns within tissues, gaining insights into spatial relationships critical for deciphering the functional organization of complex biological systems.
The emergence of single-cell sequencing and spatial transcriptomics represents a paradigm shift in NGS technologies. These innovations empower researchers to explore biological systems with unprecedented granularity, offering new dimensions of information that significantly contribute to our understanding of cellular heterogeneity and spatial relationships within tissues.
VIII. Ethical Considerations
A. Privacy and Informed Consent
- Handling sensitive genetic information: The ethical implications of NGS technologies extend to the handling of sensitive genetic information. As genomic data become more accessible, it is crucial to establish robust privacy measures to safeguard individuals’ confidential information. Researchers and healthcare professionals must prioritize secure data storage, transmission, and analysis to prevent unauthorized access or breaches.
- Ensuring ethical guidelines in research and clinical applications: The use of NGS in research and clinical settings necessitates strict adherence to ethical guidelines. Researchers must obtain informed consent from study participants, clearly communicating the purposes, potential risks, and benefits of genomic analysis. Transparency in how genetic data will be used, stored, and shared is essential to uphold the principles of autonomy and respect for individuals’ choices.
Ethical considerations in NGS underscore the need for a balance between scientific advancements and the protection of individuals’ privacy and rights. By implementing robust privacy protocols and ensuring informed consent, the ethical framework surrounding NGS can evolve in a manner that promotes trust, transparency, and responsible use of genomic information.
IX. Future Directions
A. Technological Advancements
- Improvements in sequencing accuracy and throughput: The future of NGS is likely to witness continuous advancements in sequencing technologies. Enhanced accuracy and increased throughput will empower researchers to generate more precise and comprehensive genomic data. This progress will contribute to a deeper understanding of genetic variations, enabling the identification of rare variants and improving the overall reliability of genomic analyses.
- Integration with other high-throughput technologies: The integration of NGS with other high-throughput technologies holds immense potential for multidimensional data acquisition. Combining genomic information with data from proteomics, metabolomics, and other omics fields can provide a holistic view of biological systems. This convergence of technologies will facilitate a more comprehensive understanding of complex biological processes, leading to breakthroughs in personalized medicine and systems biology.
The future directions of NGS are poised to transform genomics research and its applications, with a focus on refining existing capabilities and fostering synergies with complementary technologies. These advancements will not only drive scientific discovery but also contribute to the continued evolution of precision medicine and personalized healthcare.
X. Conclusion
A. Transformative Impact of NGS in Genomic and Genetic Research
The advent of Next-Generation Sequencing (NGS) technologies has marked a paradigm shift in genomic and genetic research. NGS has not only democratized access to genomic information but has also revolutionized the scale and scope of biological investigations. The transformative impact of NGS can be encapsulated in its ability to:
- Enable Comprehensive Genomic Profiling: NGS technologies, including Illumina, Ion Torrent, PacBio, and Nanopore sequencing, empower researchers to perform whole-genome sequencing, exome sequencing, RNA-seq, and epigenomic studies. This comprehensive genomic profiling has unveiled the intricacies of genetic information, allowing for a deeper understanding of the genome’s functional elements.
- Uncover Rare Variants and Structural Variations: NGS has facilitated the identification of rare genetic variants and structural variations, shedding light on previously elusive aspects of genetic diversity. This has significant implications for rare disease research, personalized medicine, and our understanding of the genetic underpinnings of various conditions.
- Advance Functional Genomics: With the integration of CRISPR-based functional genomics studies and high-throughput screening, NGS has propelled functional genomics to new heights. This has accelerated the annotation of gene function and provided insights into the complex regulatory networks governing cellular processes.
- Drive Precision Medicine and Pharmacogenomics: NGS plays a pivotal role in precision medicine by enabling genomic profiling in clinical settings. The ability to tailor treatment strategies based on individual genetic information and predict personalized drug responses through pharmacogenomics represents a significant stride towards more effective and targeted healthcare.
B. Shaping the Future of Biomedical Science
As we conclude, it is evident that NGS is not merely a technological advancement but a catalyst that is shaping the future of biomedical science. The ongoing improvements in sequencing accuracy, increased throughput, and the integration with other high-throughput technologies are indicative of a dynamic field that continues to evolve.
The ethical considerations surrounding privacy and informed consent underscore the responsibility associated with harnessing the power of genomic information. Striking a balance between technological advancements and ethical guidelines will be crucial as NGS continues to unfold new possibilities in research and clinical applications.
In essence, NGS stands as a cornerstone in the edifice of modern genomics. Its impact on understanding genetic variation, unraveling disease mechanisms, and paving the way for personalized approaches to healthcare is profound. As we look ahead, NGS will undoubtedly play a central role in unraveling the complexities of the genome, thereby propelling biomedical science towards new frontiers of knowledge and innovation.