Unveiling the Genomic Frontier: A Comprehensive Exploration
December 20, 2023I. Introduction
A. Overview of Genomics
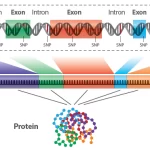
Genomics is a branch of molecular biology that focuses on the study of an organism’s complete set of DNA, known as its genome. The genome contains all the genetic information needed for the development, functioning, and regulation of an organism. Genomic research involves the analysis of DNA sequences, the identification of genes and their functions, and the exploration of genetic variations within and between species.
Key components of genomics include:
- DNA Sequencing: The process of determining the order of nucleotides in a DNA molecule. Advancements in sequencing technologies have significantly increased the speed and efficiency of genome sequencing.
- Functional Genomics: The study of gene function, including how genes are regulated, how they interact with each other, and how they contribute to cellular processes.
- Comparative Genomics: Comparing the genomes of different species to understand evolutionary relationships, identify conserved elements, and uncover the genetic basis of traits.
- Structural Genomics: Investigating the three-dimensional structure of DNA and proteins to gain insights into their functions.
B. Significance of Long-read Sequencing Technologies
- Traditional vs. Long-read Sequencing:
- Traditional Sequencing (Short Reads): In traditional sequencing methods, short fragments of DNA are sequenced, typically around 100-300 base pairs at a time.
- Long-read Sequencing: Long-read sequencing technologies, on the other hand, can read much longer stretches of DNA, often thousands to tens of thousands of base pairs in a single read.
- Advantages of Long-read Sequencing:
- Genome Assembly: Long-read sequencing aids in constructing more accurate and contiguous genome assemblies. Short-read technologies may struggle with repetitive regions, leading to fragmented assemblies, while long reads help resolve these complexities.
- Structural Variations: Long reads provide a clearer picture of structural variations in the genome, such as large insertions, deletions, inversions, and translocations. This is crucial for understanding genetic diversity and disease mechanisms.
- Transcriptomics: Long-read sequencing is valuable for studying RNA molecules, allowing for the identification of full-length transcripts, alternative splicing events, and isoform diversity.
- Applications in Genomic Research:
- Human Genomics: Long-read sequencing has implications for understanding the human genome’s structural complexity, identifying disease-associated variations, and improving the accuracy of clinical genomic analyses.
- Microbial Genomics: In microbial genomics, long-read sequencing facilitates the study of complex microbial communities, antibiotic resistance mechanisms, and the genomic plasticity of microorganisms.
- Plant and Animal Genomics: Long-read sequencing contributes to the assembly of complex plant and animal genomes, enabling researchers to explore the genetic basis of traits and adaptability.
- Emerging Technologies:
- PacBio Sequencing: Pacific Biosciences (PacBio) and its Single Molecule, Real-Time (SMRT) sequencing technology are notable contributors to long-read sequencing.
- Oxford Nanopore Sequencing: Oxford Nanopore Technologies offers portable sequencers that analyze DNA by threading it through nanopores, providing real-time, long-read sequencing capabilities.
In conclusion, long-read sequencing technologies have revolutionized genomics by overcoming limitations associated with traditional short-read sequencing methods. These advancements play a crucial role in improving genome assembly, understanding structural variations, and unraveling the complexities of diverse biological systems. The significance of long-read sequencing extends across various domains of genomics research, contributing to a more comprehensive understanding of genetic information and its functional implications.
II. Long-read Sequencing Technologies
A. Definition and Importance
Long-read sequencing technologies refer to DNA sequencing methods that generate reads significantly longer than traditional short-read sequencing methods. These technologies have become increasingly important in genomics research due to their ability to overcome challenges associated with repetitive regions, structural variations, and the assembly of complex genomes.
Importance:
- Resolution of Complex Genomic Structures: Long-read sequencing allows for the resolution of complex genomic structures, such as repetitive elements, tandem repeats, and segmental duplications, which are challenging for short-read technologies.
- Detection of Structural Variations: The ability to generate long reads facilitates the accurate detection of structural variations, including insertions, deletions, inversions, and translocations, providing a more comprehensive understanding of genome architecture.
- Improved Genome Assembly: Long-read sequencing contributes to more contiguous and accurate genome assemblies compared to short-read sequencing. This is particularly beneficial for assembling large and complex genomes with high levels of repetitive sequences.
B. Advantages in Whole Genome Assembly
- Overcoming Challenges in Repetitive Regions:
- Repetitive Sequences: Repetitive regions in the genome, such as transposons and tandem repeats, pose challenges for short-read sequencing. Long reads help span these repeats, facilitating the assembly of sequences that are difficult to resolve with short reads.
- Contiguity and Accuracy:
- Contiguous Assemblies: Long reads contribute to more contiguous genome assemblies, reducing the fragmentation often seen in short-read assemblies. This is crucial for accurately representing the organization of genes and regulatory elements.
- Reduced Ambiguity: Longer reads provide more unique sequence information, reducing ambiguity in assembly and aiding in the accurate reconstruction of genomic regions.
- Haplotype Phasing:
- Haplotype-Resolved Assemblies: Long reads are valuable for haplotype phasing, allowing the differentiation of alleles on homologous chromosomes. This is essential for understanding genetic diversity and heterozygosity within populations.
C. Applications in Studying Complex Genomic Regions
- Structural Variant Detection:
- Identification of Large Variations: Long reads enable the identification of large structural variations, including deletions, insertions, inversions, and complex rearrangements. This is crucial for understanding the genetic basis of diseases and population diversity.
- Characterization of Repeat-rich Regions:
- Repetitive Elements: Long-read sequencing aids in the characterization of repetitive elements, such as retrotransposons and satellite DNA. This is essential for studying genome evolution and understanding the functional role of repetitive sequences.
- Full-length Transcriptome Sequencing:
- Isoform Diversity: Long-read sequencing is instrumental in studying alternative splicing events and capturing full-length transcripts. This provides insights into isoform diversity and gene expression regulation, which are critical for understanding cellular functions.
- Microbial Genomics:
- Bacterial and Viral Genomes: Long-read sequencing is valuable for studying bacterial and viral genomes, especially those with high genomic plasticity. It aids in resolving complex genomic structures and understanding the dynamics of microbial communities.
In conclusion, long-read sequencing technologies play a pivotal role in genomics research by addressing the limitations of short-read sequencing methods. Their ability to generate longer reads contributes to more accurate and contiguous genome assemblies, facilitates the detection of structural variations, and enhances our understanding of complex genomic regions, ultimately advancing our knowledge of genetic diversity and functional genomics.
III. Functional Genomics
A. Defining Functional Genomics
Functional genomics is a branch of genomics that focuses on understanding the function and interactions of genes, as well as the non-coding elements that regulate gene expression. Unlike structural genomics, which primarily involves the identification and mapping of genes and their sequences, functional genomics aims to decipher the roles and relationships of genes in the context of biological processes. It employs various experimental and computational approaches to explore how genetic information is utilized and regulated to carry out the functions of living organisms.
Key components of functional genomics include:
- Gene Function Analysis: Investigating the biological roles of individual genes, understanding their contributions to cellular processes, and identifying the consequences of gene perturbations.
- Regulatory Element Exploration: Examining the non-coding regions of the genome, such as promoters, enhancers, and other regulatory elements, to understand how they control gene expression and cellular functions.
- High-throughput Technologies: Utilizing advanced technologies, such as transcriptomics, proteomics, and epigenomics, to generate large-scale data sets that provide insights into gene expression, protein interactions, and epigenetic modifications.
B. Roles of Genes and Regulatory Elements
- Gene Functions:
- Protein-Coding Genes: Investigating the functions of protein-coding genes involves understanding how the encoded proteins contribute to cellular processes, signaling pathways, and overall organismal functions.
- Non-Coding RNAs: Exploring the roles of non-coding RNAs, including microRNAs and long non-coding RNAs, in gene regulation, mRNA stability, and other cellular functions.
- Regulatory Elements:
- Promoters and Enhancers: Identifying and characterizing promoters and enhancers that control the initiation and modulation of gene transcription.
- Epigenetic Modifications: Studying epigenetic modifications, such as DNA methylation and histone modifications, to understand their impact on gene expression and cellular differentiation.
- Functional Screens:
- Genetic Screens: Conducting systematic genetic screens, such as CRISPR/Cas9 knockout screens, to identify genes that are essential for specific cellular processes or pathways.
- RNAi Screens: Using RNA interference (RNAi) to selectively silence gene expression to study gene function.
C. Implications for Health and Disease
- Disease Mechanisms:
- Identification of Disease-Associated Genes: Functional genomics helps identify genes associated with diseases and provides insights into the molecular mechanisms underlying pathological conditions.
- Pathway Analysis: Understanding how genes function within specific pathways allows researchers to unravel the complex network of interactions involved in diseases.
- Drug Discovery:
- Target Identification: Functional genomics aids in the identification of potential drug targets by revealing genes or pathways crucial for disease progression.
- Drug Response Prediction: Studying the functional genomics of individual patients may enable the prediction of drug responses and the development of personalized treatment strategies.
- Precision Medicine:
- Genomic Biomarkers: Functional genomics contributes to the discovery of genomic biomarkers associated with disease susceptibility, prognosis, and treatment response.
- Tailoring Therapies: Understanding the functional genomics of individual patients allows for the customization of therapies based on their unique genetic makeup.
- Cellular and Developmental Biology:
- Stem Cell Differentiation: Investigating gene function during stem cell differentiation provides insights into developmental processes and potential applications in regenerative medicine.
- Cell Signaling Pathways: Understanding the roles of genes in cell signaling pathways contributes to knowledge about normal cellular function and aberrations in disease.
In conclusion, functional genomics is integral to unraveling the complexity of genetic information and its impact on cellular functions. By exploring gene function and regulatory elements, functional genomics has far-reaching implications for our understanding of health, disease, drug discovery, and the development of precision medicine. It serves as a key component in deciphering the functional aspects of the genome and translating this knowledge into practical applications in biomedical research and healthcare.
IV. Non-coding DNA
A. Understanding Non-coding DNA
Non-coding DNA refers to the portions of the genome that do not encode protein sequences. While protein-coding genes represent a relatively small fraction of the genome, non-coding DNA constitutes the majority. Historically considered “junk DNA,” the functional significance of non-coding DNA has become a focal point of genomics research, revealing a diverse array of regulatory elements and functional non-coding RNAs.
Key components of non-coding DNA include:
- Promoters and Enhancers: Non-coding regions play crucial roles in the regulation of gene expression. Promoters are sequences that initiate transcription, and enhancers are regions that enhance gene expression by interacting with promoters.
- Introns: Non-coding regions within protein-coding genes, known as introns, are transcribed but not translated. They are later spliced out during RNA processing, and their functional significance is an area of active research.
- Intergenic Regions: The stretches of DNA located between genes, called intergenic regions, were initially thought to lack function. However, emerging evidence suggests that these regions may contain regulatory elements influencing nearby genes.
- Non-coding RNAs: Non-coding DNA produces various non-coding RNAs (ncRNAs), including microRNAs, long non-coding RNAs (lncRNAs), and small nuclear RNAs (snRNAs). These play roles in gene regulation, chromatin remodeling, and other cellular processes.
B. Functional Significance
- Regulation of Gene Expression:
- Promoters and Enhancers: Non-coding DNA contains promoters and enhancers that play critical roles in the regulation of gene expression. These elements control when, where, and to what extent genes are transcribed.
- Non-coding RNAs:
- MicroRNAs: MicroRNAs are short ncRNAs that post-transcriptionally regulate gene expression by binding to messenger RNAs (mRNAs), leading to their degradation or inhibition of translation.
- Long Non-coding RNAs (lncRNAs): lncRNAs participate in diverse cellular processes, acting as regulators of gene expression, chromatin modifiers, and scaffolds for protein complexes.
- Structural and Evolutionary Functions:
- Chromatin Organization: Non-coding DNA plays a role in maintaining the structural organization of chromosomes. Structural elements, such as insulators, contribute to proper chromatin folding and gene regulation.
- Evolutionary Conservation: Some non-coding regions are conserved across species, indicating their functional importance in evolution, even if their precise roles are not fully understood.
C. Exploring the Majority of the Human Genome
- Functional Genomics Approaches:
- ChIP-Seq (Chromatin Immunoprecipitation Sequencing): Identifies regions of the genome associated with specific proteins, providing insights into transcription factor binding sites and chromatin modifications.
- RNA-Seq: Examines the transcriptome, including non-coding RNAs, to understand their expression patterns and potential functions.
- CRISPR/Cas9 Screens: Genome-wide functional screens can reveal the impact of disrupting non-coding regions on cellular processes.
- ENCODE Project (Encyclopedia of DNA Elements):
- Comprehensive Annotation: The ENCODE project aims to identify all functional elements in the human genome, including non-coding regions. It provides a comprehensive annotation of regulatory elements and their roles in gene regulation.
- Technological Advances:
- Long-Read Sequencing Technologies: Advances in sequencing technologies, such as long-read sequencing, contribute to a more accurate and comprehensive understanding of non-coding regions, including their structural features and functional elements.
- Functional Annotation Databases:
- GENCODE: An example of a functional annotation database that provides information on the structure and function of protein-coding and non-coding genes in the human genome.
In conclusion, the exploration of non-coding DNA is transforming our understanding of the genome. Beyond its initial characterization as “junk DNA,” non-coding regions are now recognized for their crucial roles in gene regulation, chromatin organization, and evolutionary processes. Advances in functional genomics approaches and technologies continue to unveil the functional significance of non-coding DNA, opening new avenues for research in genomics and molecular biology.
V. Population Genomics
A. Introduction to Population Genomics
Population genomics is a field of study that focuses on the analysis of genetic variation within and between populations. It involves the application of genomic techniques to investigate the genetic composition, evolutionary history, and dynamics of populations over time. By studying the genetic diversity within and among groups of individuals, population genomics provides insights into the forces shaping genetic variation, including natural selection, migration, and demographic events.
Key aspects of population genomics include:
- Genetic Variation: Examining the presence of genetic variations, such as single nucleotide polymorphisms (SNPs), insertions, deletions, and structural variations, across populations.
- Evolutionary Processes: Investigating the impact of evolutionary processes, such as natural selection, genetic drift, and gene flow, on the distribution of genetic variation within and between populations.
- Demographic History: Analyzing the demographic history of populations, including population size changes, migration events, and bottlenecks, to understand patterns of genetic diversity.
- Adaptation: Studying the genetic basis of adaptation to different environments, including responses to selective pressures and environmental challenges.
B. Investigating Genetic Diversity
- Genomic Variation:
- Single Nucleotide Polymorphisms (SNPs): SNPs are common genetic variations where a single nucleotide differs among individuals. Population genomics analyzes the distribution of SNPs to understand the genetic landscape.
- Copy Number Variations (CNVs): CNVs involve variations in the number of copies of DNA segments. Population genomics explores the prevalence and impact of CNVs on genetic diversity.
- Genome-Wide Association Studies (GWAS):
- Identifying Genetic Associations: GWAS analyze genetic variations across the entire genome to identify associations between specific genetic variants and traits or diseases within populations.
- Haplotypes and Linkage Disequilibrium:
- Haplotype Analysis: Haplotypes are combinations of alleles on a single chromosome. Population genomics studies haplotype structure and diversity to understand the inheritance patterns of genetic variants.
- Linkage Disequilibrium (LD): LD measures the non-random association of alleles at different loci. Population genomics investigates LD patterns to understand the genetic architecture of populations.
C. Role in Disease Susceptibility and Adaptation
- Disease Susceptibility:
- Common Disease Variants: Population genomics helps identify common genetic variants associated with susceptibility to diseases. This knowledge is crucial for understanding disease risk and developing personalized medicine approaches.
- Population-Specific Variants: Some disease-associated variants may be specific to certain populations, highlighting the importance of considering genetic diversity in healthcare research.
- Adaptation to Environments:
- Local Adaptation: Populations may undergo genetic changes in response to local environmental conditions, such as climate, altitude, or pathogen exposure. Population genomics helps identify candidate genes involved in adaptation.
- Selective Sweeps: Analysis of selective sweeps, where advantageous alleles rapidly increase in frequency, provides insights into the genetic basis of adaptation.
- Pharmacogenomics:
- Population-Specific Drug Responses: Population genomics contributes to pharmacogenomics by identifying genetic variants associated with variations in drug response among populations. This information guides personalized medicine approaches.
- Conservation Genetics:
- Endangered Populations: In conservation genetics, population genomics is used to assess the genetic diversity and health of endangered populations. This information informs conservation strategies to maintain genetic viability.
- Human Migration History:
- Tracing Ancestry and Migration Patterns: Population genomics contributes to understanding human migration patterns, historical demography, and the peopling of different regions.
In conclusion, population genomics provides a comprehensive view of genetic diversity, evolutionary processes, and the genetic basis of adaptation within and between populations. The field has broad implications for understanding disease susceptibility, pharmacogenomics, conservation biology, and unraveling the complex interplay between genes and environments across diverse human populations and other species.
VI. Synthesis and Future Directions
A. Integrating Insights from Long-read Sequencing, Functional Genomics, and Population Genomics
- Holistic Genomic Understanding:
- Long-Read Sequencing and Functional Genomics: Integrating insights from long-read sequencing and functional genomics enhances our understanding of the structural and functional aspects of the genome. Long reads contribute to accurate genome assembly, revealing complex genomic structures, while functional genomics uncovers the roles of genes and regulatory elements.
- Population Genomics Integration: Incorporating population genomics adds a population-level perspective, allowing researchers to explore how genetic variations and functional elements contribute to diversity within and between populations. This holistic approach facilitates a more comprehensive genomic understanding.
- Precision Medicine Applications:
- Identification of Functional Variants: Integrating data from long-read sequencing and functional genomics enables the identification of functional variants associated with disease susceptibility or drug response. This information is crucial for tailoring precision medicine approaches to individual patients based on their unique genomic profiles.
- Disease Mechanism Exploration:
- Linking Structural Variations to Function: Connecting insights from long-read sequencing on structural variations with functional genomics data helps unravel the impact of these variations on gene regulation and disease mechanisms. This integrated approach contributes to a more nuanced understanding of complex genomic disorders.
B. Potential Applications in Medicine and Research
- Advanced Diagnostics:
- Genomic Biomarkers: The integration of long-read sequencing, functional genomics, and population genomics contributes to the discovery of genomic biomarkers associated with diseases. These biomarkers have the potential to revolutionize diagnostics by providing more accurate and comprehensive information.
- Therapeutic Target Identification:
- Precision Drug Development: Understanding the functional genomics of disease and incorporating insights from long-read sequencing can aid in the identification of precise therapeutic targets. This approach is essential for developing targeted therapies with improved efficacy and reduced side effects.
- Population-Specific Medicine:
- Tailoring Treatments to Populations: Population genomics, combined with functional genomics, facilitates the identification of population-specific genetic variations that influence drug response and disease susceptibility. This knowledge supports the development of region-specific medical interventions.
- Environmental Adaptation:
- Understanding Genetic Responses to Environment: Integrating population genomics with functional genomics allows for a deeper exploration of how populations adapt to diverse environments. This knowledge can be harnessed to address health disparities and design interventions that consider genetic and environmental factors.
C. Future Trends and Developments
- Multi-Omics Integration:
- Comprehensive Data Integration: The future will likely see increased efforts to integrate data from multiple omics levels, including genomics, transcriptomics, proteomics, and epigenomics. This holistic approach provides a more complete picture of biological systems.
- Advancements in Sequencing Technologies:
- Continued Improvements in Long-Read Sequencing: Ongoing advancements in long-read sequencing technologies are anticipated, with improvements in read lengths, throughput, and cost-effectiveness. This will further enhance the accuracy of genome assemblies and the detection of complex genomic variations.
- Functional Annotation and Interpretation:
- Advances in Functional Genomics Tools: The development of advanced tools for functional genomics, including CRISPR-based technologies and high-throughput functional assays, will enable more precise annotation and interpretation of genomic elements. This will aid in uncovering the functional consequences of genetic variations.
- Population Genomics on a Global Scale:
- Diverse Population Representation: Future population genomics research is expected to emphasize broader representation of diverse populations globally. This approach will contribute to a more inclusive understanding of genetic diversity and disease susceptibility across different ethnic groups.
- Ethical and Social Considerations:
- Ethical Genomic Research Practices: As genomic research advances, there will be a heightened focus on ethical considerations, data privacy, and responsible research practices. Ensuring equitable access to benefits and addressing potential societal implications will be integral to the future of genomics.
In conclusion, the synthesis of insights from long-read sequencing, functional genomics, and population genomics holds great promise for advancing medicine and research. The integration of these approaches contributes to a more comprehensive understanding of the genome, paving the way for precision medicine applications, targeted therapies, and a deeper exploration of genetic diversity and adaptation. Ongoing advancements in sequencing technologies, functional genomics tools, and ethical research practices will shape the future landscape of genomics research and its applications in healthcare and beyond.
VII. Conclusion
A. Recap of Key Findings
- Long-Read Sequencing Advances:
- Long-read sequencing technologies have revolutionized genomics, enabling more accurate genome assemblies, especially in regions with complex structures and repetitive elements.
- Functional Genomics Insights:
- Functional genomics has unveiled the roles of non-coding DNA, regulatory elements, and diverse RNA molecules in gene expression, cellular processes, and disease mechanisms.
- Population Genomics Dynamics:
- Population genomics has provided a comprehensive understanding of genetic diversity, evolutionary processes, and the genetic basis of disease susceptibility within and between populations.
- Integration for Holistic Understanding:
- Integrating long-read sequencing, functional genomics, and population genomics offers a holistic approach, providing a deeper and more nuanced understanding of the genome at structural, functional, and population levels.
B. Importance of Genomic Research in Advancing Science
- Transformative Impact on Medicine:
- Genomic research has significantly impacted medicine by contributing to the identification of disease biomarkers, the understanding of disease mechanisms, and the development of personalized and precision medicine approaches.
- Biological Insights and Innovation:
- Genomic research has provided crucial insights into the fundamental processes of life, including gene regulation, cellular functions, and evolution. These insights have paved the way for innovative applications in diverse fields.
- Disease Prevention and Treatment:
- Advances in genomics have enhanced our ability to prevent, diagnose, and treat diseases. Genomic information is increasingly integrated into clinical practice, guiding therapeutic decisions and improving patient outcomes.
- Precision and Personalized Approaches:
- Genomic research has facilitated the shift towards precision and personalized approaches in healthcare. Tailoring treatments to individual genetic profiles has the potential to improve treatment efficacy and reduce adverse effects.
C. Call to Action: Continuing the Exploration of Genomic Frontiers
- Investment in Research and Technology:
- Continued investment in genomics research and technological developments is essential. This includes supporting advancements in sequencing technologies, functional genomics tools, and computational methods to handle vast genomic datasets.
- Ethical Considerations and Diversity:
- Researchers and policymakers must prioritize ethical considerations, ensuring responsible data use, protecting privacy, and addressing potential disparities in the application of genomic knowledge across diverse populations.
- Education and Public Awareness:
- Educating the public and healthcare professionals about genomics is crucial. Promoting genomic literacy fosters informed decision-making, encourages participation in research, and enhances the integration of genomics into healthcare practices.
- Interdisciplinary Collaboration:
- Encouraging collaboration across disciplines, including biology, medicine, computer science, and ethics, fosters a comprehensive and integrative approach to genomic research. Cross-disciplinary collaboration accelerates discoveries and applications.
- Global Collaboration in Population Genomics:
- Enhancing global collaboration in population genomics research ensures the inclusion of diverse populations and contributes to a more comprehensive understanding of genetic diversity, evolution, and disease susceptibility on a global scale.
In conclusion, the exploration of genomic frontiers has transformed our understanding of life at the molecular level. The integration of long-read sequencing, functional genomics, and population genomics provides a multifaceted view of the genome, with far-reaching implications for medicine, biology, and society. As we stand at the intersection of technological innovation and scientific discovery, a continued commitment to genomic research and responsible application is vital to unlock the full potential of the genomic revolution and shape the future of healthcare and biological sciences.